- IT
- |
- EN
The use of antiseptics and disinfectants represents one of the fundamental principles of hygiene and is increasingly linked to the global strategy for the reduction of healthcare-associated infections (HAIs) and, consequently, to patient and healthcare worker safety [1].
The chemical substances used for antisepsis and disinfection exert a microbicidal action against infectious agents by inhibiting, eliminating, or destroying pathogenic microorganisms. However, several factors may influence their effectiveness, including microbial load, intended use, solution concentration, contact time, temperature and pH, intrinsic microbial resistance, the presence of organic material, and biofilm [2,3].
For this reason, procedures for the use of antiseptics and disinfectants adopted by healthcare professionals must be: consistent with the information provided in technical data sheets; based on the main recommendations [4]; and appropriate in the selection of the substance according to its intended use.
These measures represent crucial aspects for preventing the risk of product contamination [5], which in turn may be responsible for outbreaks in healthcare settings [6], and especially for counteracting the emerging phenomenon of resistance selection to certain antiseptics and disinfectants, as well as cross-resistance between antibiotics and disinfectants (antimicrobial resistance) [7].
It is therefore necessary to use products that demonstrate proven efficacy [8], according to specific UNI EN technical standards [9], for the required antimicrobial activity (virucidal, bactericidal, sporicidal) [10]. This must also consider the required contact times, the different substrates, and possible interfering substances in which infectious agents may be present, since microbicidal properties could otherwise be insufficient, neutralized, or significantly reduced [11].
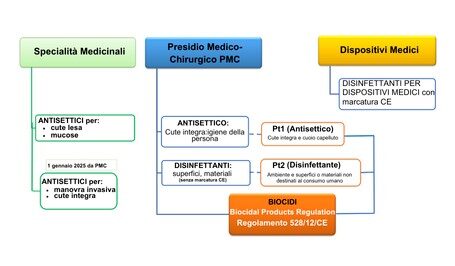
According to the regulatory framework governing market authorization (Scheme 1), antiseptics and disinfectants are classified as follows:
Medicinal products (pharmaceutical specialties) – antiseptics intended for damaged skin and mucous membranes. According to the Decree of the Italian Ministry of Health of 29 March 2023, which entered into force on 1 January 2025, antiseptics used on intact skin prior to a medical treatment (e.g., invasive procedures) are also classified as medicinal products.
Medical and Surgical Presidia (PMC) or Biocides – antiseptics for intact skin or personal hygiene (Product Type 1 – PT1) and disinfectants for surfaces and materials without CE marking (Product Type 2 – PT2), in accordance with EU Regulation 528/2012, aimed at ensuring a high level of protection for human health and the environment.
Medical Devices – only for materials with CE marking [4].
Biocide: any substance or mixture, in the form supplied to the user, consisting of or capable of generating one or more active substances, intended to destroy, deter, render harmless, prevent the action of, or otherwise control harmful organisms, by any means other than mere physical or mechanical action (Article 3 of EU Regulation 528/2012).
Disinfection activity: the set of procedures and operations aimed at disinfecting confined environments, related areas, and surfaces, through the destruction or inactivation of pathogenic microorganisms (Italian National Institute of Health – ISS Report No. 19) [5].
Antisepsis: a process aimed at the elimination or inactivation of pathogenic microorganisms present on skin and mucous membranes.
Disinfection: a process by which most microorganisms, but not bacterial spores, are eliminated or destroyed from inanimate objects or surfaces.
Cleaning: the removal of foreign material (dirt, organic matter, etc.) from objects, surfaces, skin, and mucous membranes.
Detergent: Article 2.1 of Regulation (EC) No. 648/2004 defines a detergent as “any substance or mixture containing soaps and/or other surfactants intended for washing and cleaning processes” (ISS Report No. 19) [5].
The cleaning and/or washing procedure reduces the number of microorganisms, removes organic material, and facilitates the penetration of the active substance. It must therefore precede antisepsis and disinfection procedures.
[1] Rutala WA, Boyce JM, Weber DJ. Disinfection, sterilization and antisepsis: An overview. Am J Infect Control. 2023 Nov;51(11S):A3-A12. doi: 10.1016/j.ajic.2023.01.001. Review. PubMed .
[2] ANMDO. I quaderni di Direzione Sanitaria. Disinfezione e Antisepsi. Manuale per il corretto utilizzo degli antisettici e disinfettanti. Edizione 2025.
[3] SIFO. Disinfettanti e Antisettici dalla norma al prontuario galenico. 2024.
[4] ANMDO. Linee guida per il corretto utilizzo degli antisettici - disinfettanti. 2009
[5] Lompo, P.; Heroes, A.-S.; Agbobli, E.; Kühne, V.; Tinto, H.; Affolabi, D.; Jacobs, J. Bacterial Contamination of Antiseptics, Disinfectants and Hand Hygiene Products in Healthcare Facilities in High-Income Countries: A Scoping Review. Hygiene 2023, 3, 136-175.
[6] Weber DJ, Rutala WA, Sickbert-Bennett EE. Outbreaks associated with contaminated antiseptics and disinfectants. Antimicrob Agents Chemother. 2007
[7] Rozman U, Pušnik M, Kmetec S, Duh D, Šostar Turk S. Reduced Susceptibility and Increased Resistance of Bacteria against Disinfectants: A Systematic Review. Microorganisms. 2021
[8] Rutala W.A, Weber D.J. Selection of the Ideal Disinfectant. Infect Control Hosp Epidemiol. 2014
[9] UNI EN UNI EN 14885:2019. Disinfettanti chimici ed antisettici - Applicazione delle Norme Europee per idisinfettanti chimici e gli antisettici ·
[10] ECHA – European Chemical Angency, Guidance on the Biocidal Products Regulation, version 5.0 November 2022
[11] Rapporto ISS COVID-19 n. 19/2020 Rev.- Raccomandazioni ad interim sui disinfettanti nell’attuale emergenza COVID-19: presidi medico-chirurgici e biocidi. Versione del 13 luglio 2020
Specialità medicinale: Decreto L.vo n.178 del 29 maggio 1991. -Decreto L. n.219 del 24 aprile 2006 in attuazione della Direttiva 2001/83/CE e Decreto Direttoriale del Ministero della Salute del 29/03/2023
PMC: DPR n.392/1998 del 6 ottobre 1998 e Provvedimento del 5 febbraio 1999, che disciplina i PMC.
Biocida: Direttiva 98/8/CE sui biocidi (recepita Italia con DLvo n.174/2000) e Reg. (Ue) 528/2012 del 22 maggio 2012 e attuata in Italia con il nuovo Regolamento a partire dal 1° settembre 2013 - DM: Regolamento 2017/745 MDR (30;39;40;41), recentemente subentrato alla Direttiva CEE 93/42, poi recepita dal D. L.vo n.46 del 24/02/97 (“tutti i dispositivi che sono destinati specificatamente ad essere usati per disinfettare i DM)
Indicazioni strategico-operative per l’adozione della soluzione disinfettante come specialità medicinale per l’antisepsi della cute integra prima di un trattamento medico-chirurgico.
Istruzione Operativa allestimento soluzione CLOREXIDINA DIGLUCONATO AL 2% IN ALCOL AL 70% MULTIDOSE NON STERILE
[Last update February2025]
Content and updates curated by Maria Rosaria Esposito